|
1/29/2024 0 Comments Body fluid compartments powerpoint
Principle of Osmosis Osmosis is the net movement of water molecules through a selectively permeable membrane During osmosis water molecules pass through a selectively permeable membrane in two ways (1) through the lipid bilayer and (2) through aquaporins (aqua = water) channels. A solution that has 1 osmole of solute dissolved in 1 liter (1000 ml) of water is said to have an Osmolarityof 1 osmole per liter.A solution that has 1 osmole of solute dissolved in 1 Kilogram of water is said to have an Osmolalityof 1 osmole per kilogram.Therefore, 1 gram molecular weight of NaCl (58.5 gm) is equal to 2 osmole because NaCl dissociates into Na+ and Cl- (and both are osmotically active).Thus, 180 grams of glucose, which is 1 gram molecular weight of glucose, is equal to 1 osmole of glucose because glucose does not dissociate.One osmole is 1 gram molecular weight of undissociated solute.Regulation of water and solute loss This is mainly done by the kidneys Regulation of body water gain Baroreceptor 2% decrease in body mass due to fluid loss causes mild dehydration Rate of formation of metabolic water is not regulated to maintain homeostasis Water gain is regulated mainly by drinking through thirst mechanism 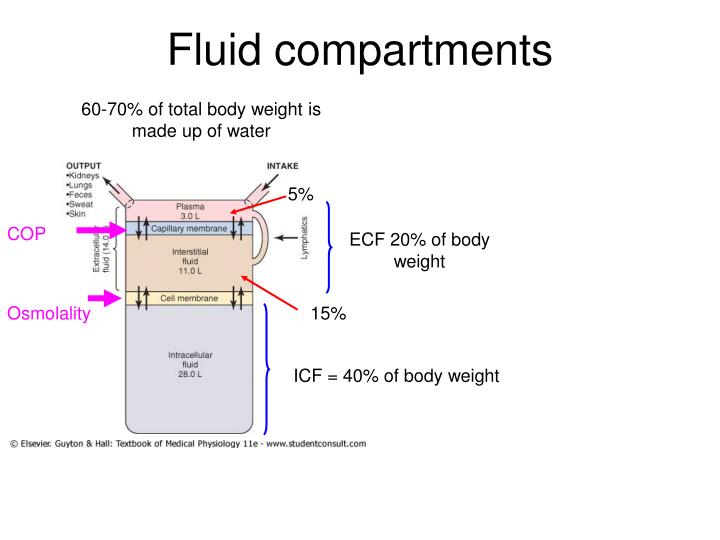
Sourse of metabolic water Rate of formation of metabolic water is not regulated Sources of body water gain and loss Body fluid volume remains constant because water loss is equal to water gain 
The osmosis, diffusion, filtration and reabsoption provide for continual exchange of water and solutes (electrolytes) among different body compartments Yet, the volume of the fluid in different body compartments remains remarkably stableĭynamics of capillary exchange (Starling’s Law of capillaries)Ĭoncentration of important solutes in ICF, ECF Body fluid compartment ECF also includes lymph, CSF, synovial fluid, aqueous & vitreous humor, endolymph & perilymph and fuild present in pleural, pericardial and peritoneal cavities
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
